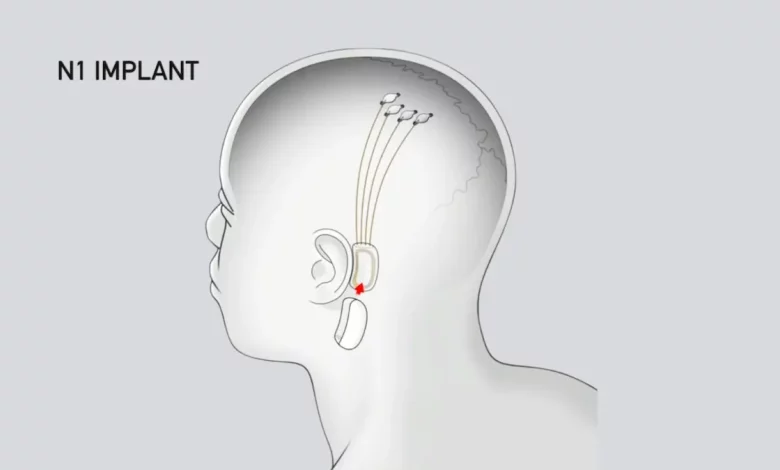
Elon Musk’s neurotech startup Neuralink announced on Thursday that it has secured approval from the Food and Drug Administration (FDA) to conduct its first in-human clinical trial, marking a significant milestone for the groundbreaking technology.
Neuralink is in the process of developing a brain implant known as the N1 Link, designed to enable patients with severe paralysis to control external technologies using neural signals alone. The potential implications of this technology are profound, with the possibility that individuals suffering from degenerative diseases such as ALS could regain their ability to communicate by moving cursors and typing using their minds.
This approval comes after the company faced several hurdles, including a rejected FDA application due to concerns about the lithium battery of the device, the possibility of the implant’s wires migrating within the brain, and the challenge of safely extracting the device without damaging brain tissue.
Despite these obstacles, Neuralink has demonstrated significant progress, as evidenced by successful implantation tests conducted on monkeys. These tests enabled the animals to manipulate cursors and play video games through the Neuralink interface, underlining the potential of this technology to facilitate direct brain-computer communication.
The FDA approval is a testament to the “incredible work by the Neuralink team in close collaboration with the FDA”, according to the company’s official tweet. While recruitment for the clinical trial is not yet open, Neuralink promises to provide more information soon.
Make tech-ish your favourite news source
Star tech-ish.com on Google. We move up your daily feed.
Elon Musk, co-founder of Neuralink, envisions a future where brain implants could cure a range of conditions including obesity, autism, depression, and schizophrenia. Moreover, he believes these devices could enable web browsing and telepathy. Late last year, Musk even expressed his confidence in the devices’ safety, stating he would be willing to implant them in his children.
The potential applications of Neuralink’s technology extend beyond the medical field. Musk has expressed concern over the potential dominance of artificial intelligence and aims to enhance human intellectual capabilities through brain-machine interfaces. He sees the potential for restoring lost capabilities, such as vision and mobility, in humans who have experienced impairments.
The journey to this point has not been without controversy. Neuralink has been the subject of several federal probes, including investigations into potential animal-welfare violations and concerns regarding the handling of dangerous pathogens. Despite these challenges, Neuralink has remained committed to its mission and continues to make strides toward its ambitious goals.
Neuralink’s achievement marks an important step in the emerging brain-computer interface (BCI) industry and a promising leap towards a future where human cognition and technology are seamlessly integrated. As the world waits for further details about the upcoming clinical trial, Neuralink’s journey continues to be a fascinating watch.
Neuralink was co-founded by Elon Musk in 2016 with the goal of developing ultra-high bandwidth brain-machine interfaces to connect humans and computers. Its mission is to enable humans to merge with AI, helping humanity avoid a potential future where AI outpaces human intelligence.








